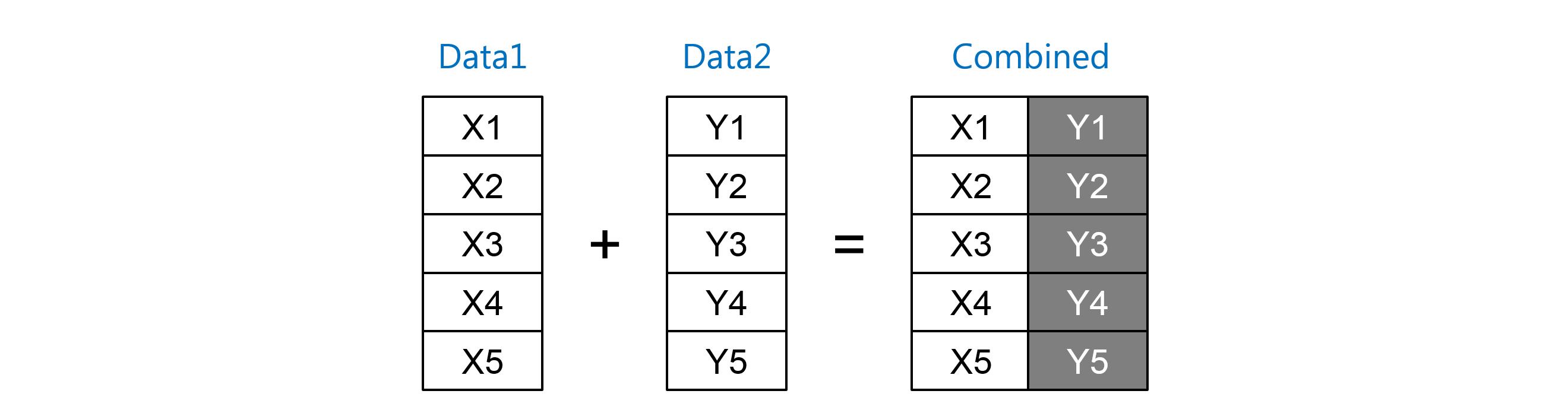
- #Basic clinical sas interview questions and answers pdf pdf#
- #Basic clinical sas interview questions and answers pdf trial#
- #Basic clinical sas interview questions and answers pdf download#
This helps keeping the size of VAD to minimum and improves the performance as well. One important thing to keep is mind while generating VAD is to make sure to exclude the variables which will never be used for any kind of analysis and are normally collected for the data collection facilitation. Sometimes the analysis datasets will have the variables not necessarily required to generate the statistical reports but sometimes they may required generating the ad-hoc reports. The safety as well as efficacy endpoints (parameters) dictate the type of the datasets are required by the clinical study for generating the statistical reports of the TLG’s. Variables, which are derived for the raw data, are used to produce the TLG’s of the clinical study. Analysis datasets contains the raw data and the variables derived from the raw datasets. DM (Demographics), MH (Medical History), AE (Adverse Events), PE (Physical Examination), EG (ECG), VS (Vital Signs), CM (Concomitant Medication), LB (Laboratory), QS (Questionnaire), IE (Inclusion and Exclusion), ), DT (Death), CO (Comments), EX (Exposure), Final, Primdiag, Pcancer, Prad, Bpbp How did you create analyzed data sets? Analysis datasets are used for the statistical analysis of the data. 20-30 datasets is required for a study to get analyzed for the safety and efficacy parameters. How many analyzed data sets did you create? Again it depends on the study the safety and efficacy parameters that are need to determined from the study. of or relating to the kidneys What is the primary and secondary end point in your last project? RCC is Kidney Neoplasms Carcinoma Renal Cell Cancer. Failure of prior sorafinib based therapy Some of the exclusion criteria were 1. having RCC with metastases and nephrectomy 2. Some of the secondary endpoints of this study involves measurement of PFS, DR, FKSI (Cancer Related Symptoms) QOL questionnaire, Safety Profile of AG-13736įunctional Assesement of Cancer Therapy (FACT)įKSI-15: is for patients with Kidney Cancer, 15 item scale Some of the inclusion Criteria of the study were 18 or older, any gender, 1.

The primary endpoint for this study is Objective Response Rate (ORR), CR, PR by RECIST (Response Evaluation Criteria In Solid Tumors) and Response rate of Axitinib in patients with RCC. It is a Phase II, Open Label, Non Randomised, Single Group study for the findings of Safety and Efficacy of this drug.
#Basic clinical sas interview questions and answers pdf pdf#
#Basic clinical sas interview questions and answers pdf trial#
Creating and deriving the analysis datasets, listings and summary tables for different clinical trial protocols.
#Basic clinical sas interview questions and answers pdf download#
Extracting the data from various internal and external database (Oracle clinical, CSV files, Excel spreadsheets) using SAS/ACCESS, SAS/INPUT, Proc Download.What are your responsibilities? Some of them include not necessarily all of them…. ADVANCED NON-SMALL CELL LUNG CANCER But I had worked earlier on some of the Neurosciences and Specialty care like Diabetes and Depression studies. What is the therapeutic area you worked earlier? Recently I am working on Oncology therapeutic area on different cancer types like 1.
